 /molecular-3d-composition-of-amino-acid-arginine-157693924-58fdf61e3df78ca159b2591b.jpg)  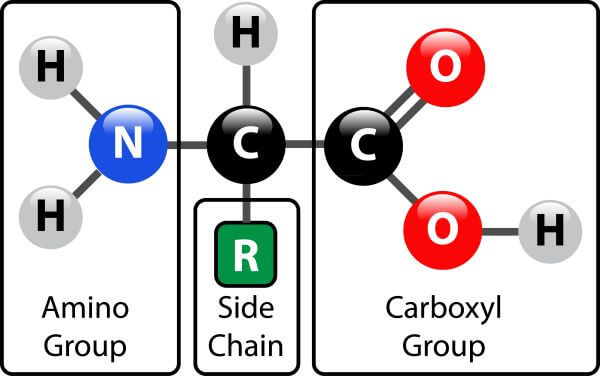
With the exception of proline, they are all 1º-amines and with the exception of glycine, they are all chiral. Some common features of these amino acids should be noted. This system is actually the only practical way of unambiguously representing the stereochemistry of complicated molecules with several asymmetric centres, but it is inconvenient with regular series of molecules like amino acids and simple sugars. The explanation given above of why the (R,S) system is little used in biochemistry is thus almost the exact opposite of reality. As mentioned, chemists also use D and L when they are appropriate to their needs. It follows that it is not just conservatism or failure to understand the (R,S) system that causes biochemists to continue with D and L: it is just that the DL system fulfils their needs much better. Worse problems can sometimes arise in substitution reactions: sometimes inversion of configuration can result in no change in the (R) or (S) prefix and sometimes retention of configuration can result in a change of prefix. However, they do not all have the same configuration in the (R,S) system: L-cysteine is also (R)-cysteine, but all the other L-amino acids are (S), but this just reflects the human decision to give a sulphur atom higher priority than a carbon atom, and does not reflect a real difference in configuration. For example, as we have seen, all of the common amino acids are L, because they all have exactly the same structure, including the position of the R group if we just write the R group as R. These rules sometimes lead to absurd results when they are applied to biochemical molecules. "In addition, however, chemists often need to define a configuration unambiguously in the absence of any reference compound, and for this purpose the alternative (R,S) system is ideal, as it uses priority rules to specify configurations. Why do biochemists still use D and L for sugars and amino acids? This explanation (taken from the link below) seems reasonable. You can remember this with the anagram CORN. As the diagram below shows, the absolute configuration of the amino acids can be shown with the H pointed to the rear, the COOH groups pointing out to the left, the R group to the right, and the NH 3 group upwards. Most naturally occurring chiral amino acids are S, with the exception of cysteine. The absolute stereochemistry is related to L-glyceraldehyde, as was the case for triacylglycerides and phospholipids. All naturally occuring proteins from all living organisms consist of L amino acids. As with lipids, biochemists use the L and D nomenclature. The amino acids are all chiral, with the exception of glycine, whose side chain is H. Hence the diversity of possible proteins is enormous. Compare this to just 65536 different oligonucleotides of 8 monomeric units (8mer). Just for an octapeptide, there are over 25 billion different possible arrangements of amino acids. Both the sequence of a protein and its total length differentiate one protein from another. In contrast, nucleic acids are polymers of just 4 different monomeric nucleotides. (Note: above picture represents the amino acid in an unlikely protonation state with the weak acid protonated and the weak base deprotonated for simplicity in showing removal of water on peptide bond formation and the hydrolysis reaction.) Proteins are polymers of twenty naturally occurring amino acids. There are many different ways to represent the structure of a polypeptide or protein, each showing differing amounts of information.įigure: Different Representations of a Polypeptide (Heptapeptide)įigure: Amino Acids React to Form Proteins At some point, when the structure is long enough, it is called a protein. Likewise, we can have tripeptides, tetrapeptides, and other polypeptides. When two amino acids link together to form an amide link, the resulting structure is called a dipeptide. Structure and Property of the Naturally-Occurring Amino Acids (Too large to include in text: print separately).In a reverse reaction, the peptide bond can be cleaved by water (hydrolysis). (We will discuss this activation by ATP later in the course.) The resulting link between the amino acids is an amide link which biochemists call a peptide bond. The carboxyl group of the amino acid must first be activated to provide a better leaving group than OH. \)Īmino acids form polymers through a nucleophilic attack by the amino group of an amino acid at the electrophilic carbonyl carbon of the carboxyl group of another amino acid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
